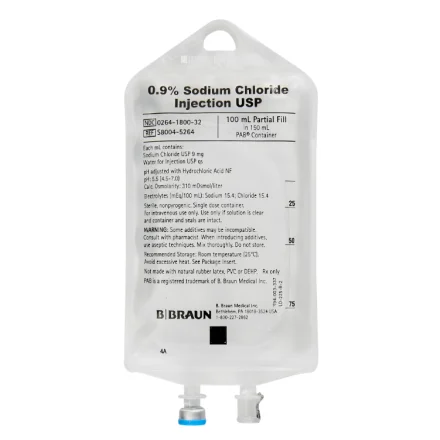
B Braun S8004-5264 Injection Solution Sod Cl.9% 100/150mL Non-DEHP/Non-PVC 100ml, 64 EA/CA
$329.34 / Parcel
Expected Price
Sodium Chloride Injections USP are sterile, nonpyrogenic, isotonic and contain no bacteriostatic or antimicrobial agents
These intravenous solutions are indicated for use in adults and pediatric patients as sources of electrolytes and water for hydration
The PAB® container is PVC-Free and DEHP-Free
The plastic container is a copolymer of ethylene and propylene formulated and developed for parenteral drugs
The copolymer contains no plasticizers and exhibits virtually no leachability
The container has two ports, one is for the intravenous administration set and the other is a medication addition site
Each is covered by a tamperproof barrier
This product is designed for use as a diluent and delivery system for intermittent intravenous administration of compatible drug additives
Support Payment Term:

HSBC Hong Kong

paypal

Alibaba Pay

Western Union

USD

EUR

GBP

SGD

HKD

CNH

CAD

MXN

BRL

JPY

THB

MOP

AUD

NZD

PLN

CZK

HUF

RON

CHF

SEK

NOK

DKK

TRY

AED

SAR

ILS

ZAR
$0
Quantity
Discover similar items
Page 1 of 2
Sodium Chloride Injections USP are sterile, nonpyrogenic, isotonic and contain no bacteriostatic or antimicrobial agents
These intravenous solutions are indicated for use in adults and pediatric patients as sources of electrolytes and water for hydration
The PAB® container is PVC-Free and DEHP-Free
The plastic container is a copolymer of ethylene and propylene formulated and developed for parenteral drugs
The copolymer contains no plasticizers and exhibits virtually no leachability
The container has two ports, one is for the intravenous administration set and the other is a medication addition site
Each is covered by a tamperproof barrier
This product is designed for use as a diluent and delivery system for intermittent intravenous administration of compatible drug additives